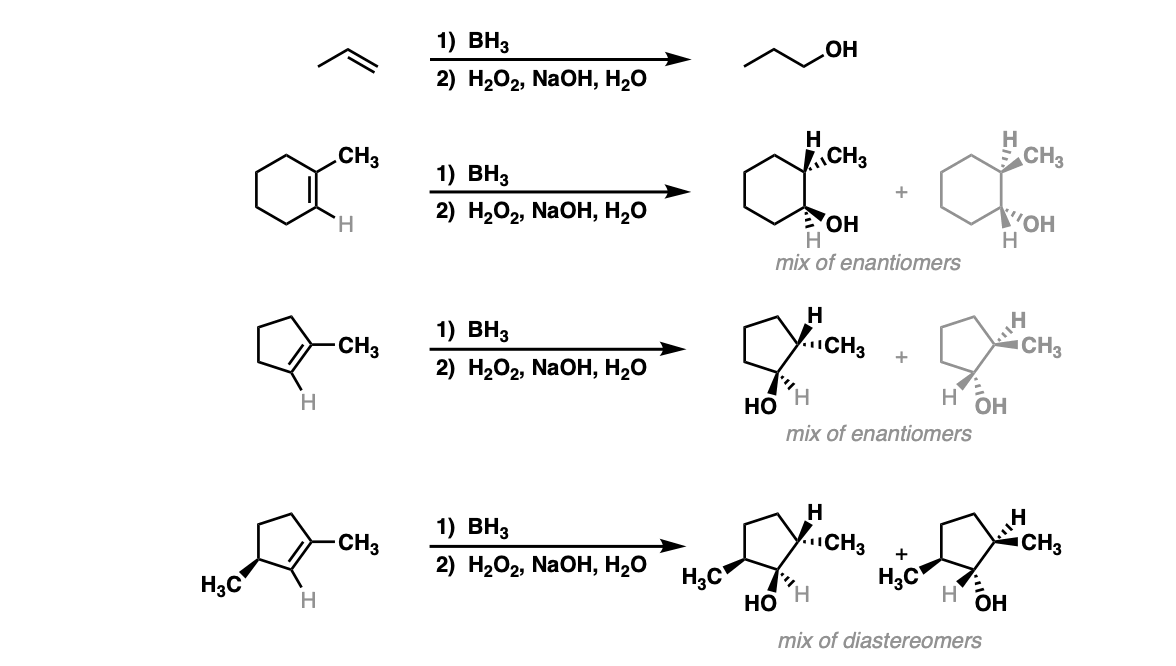
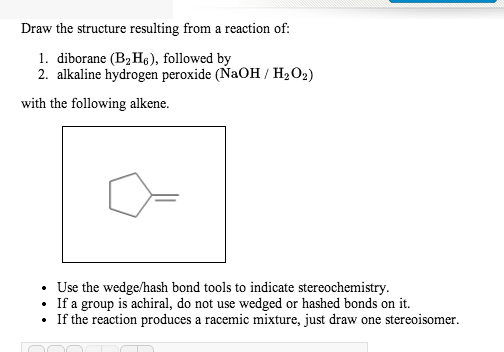
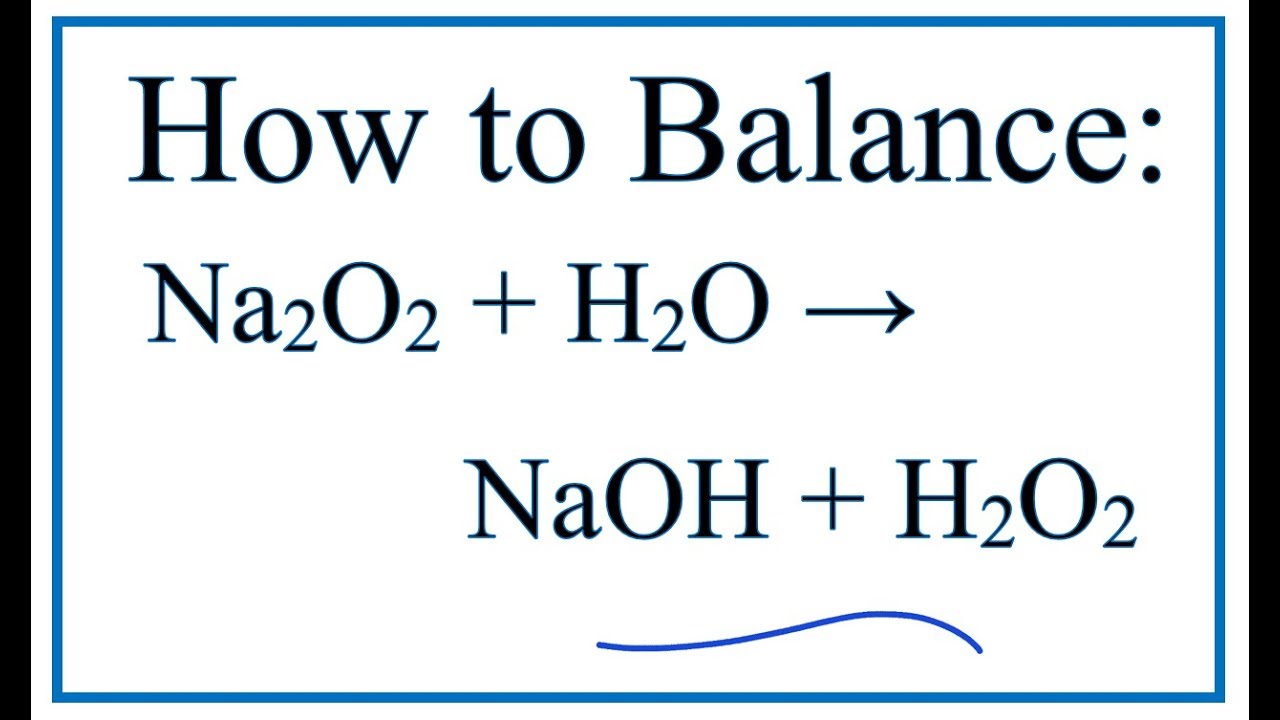
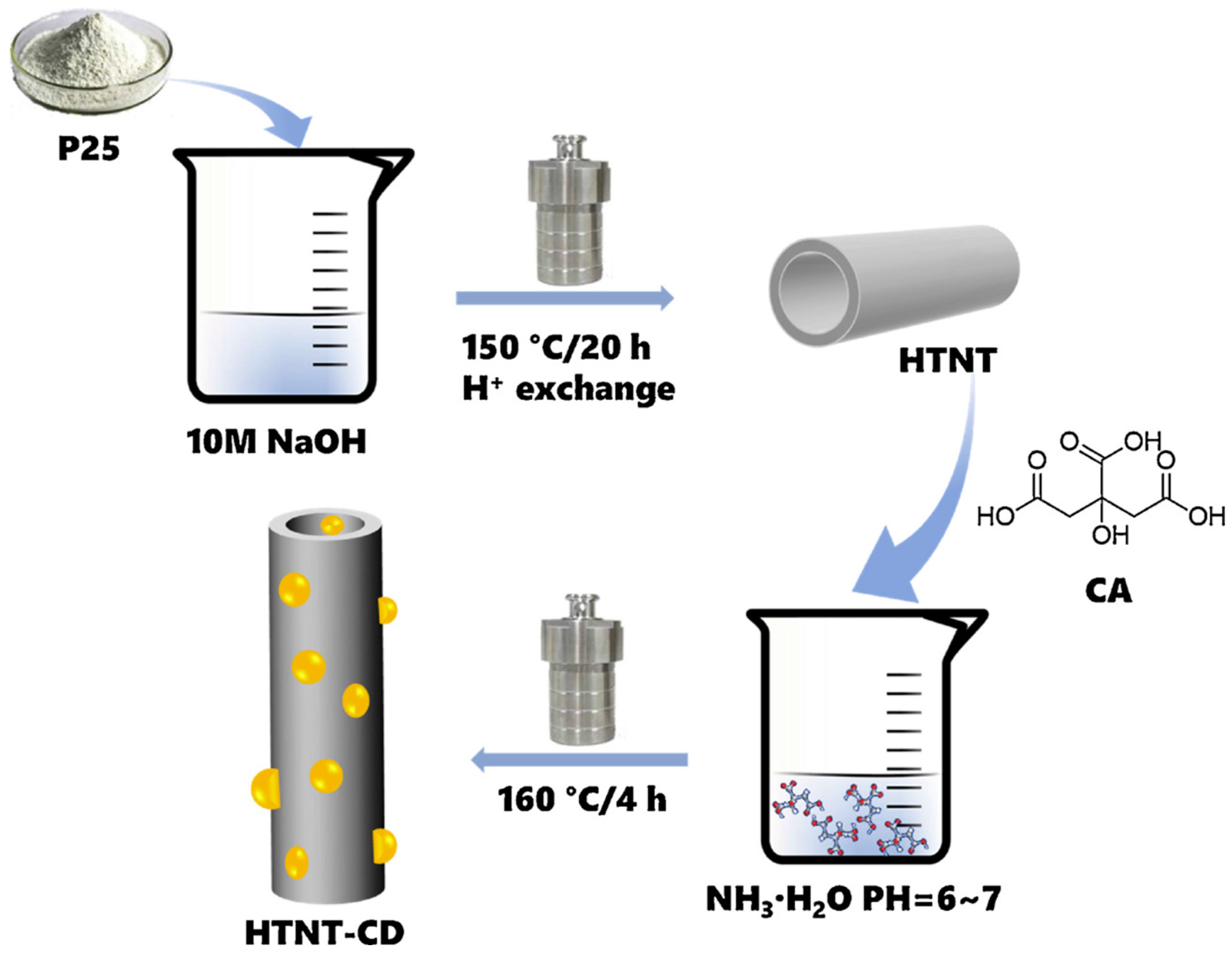
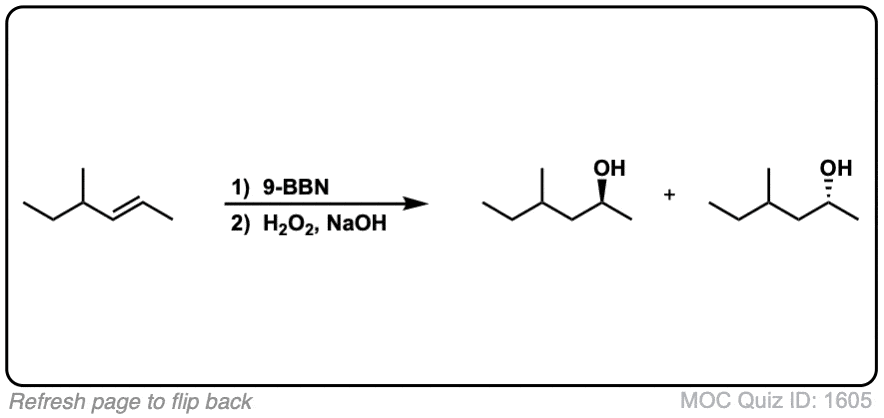
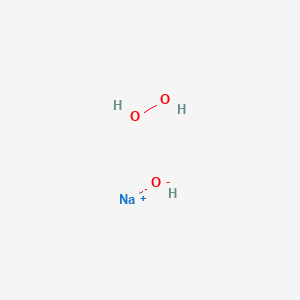
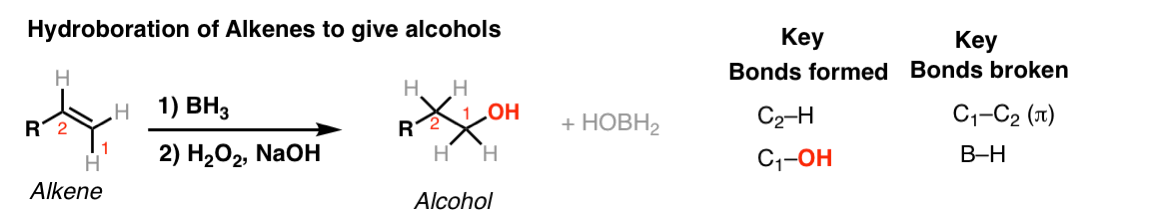
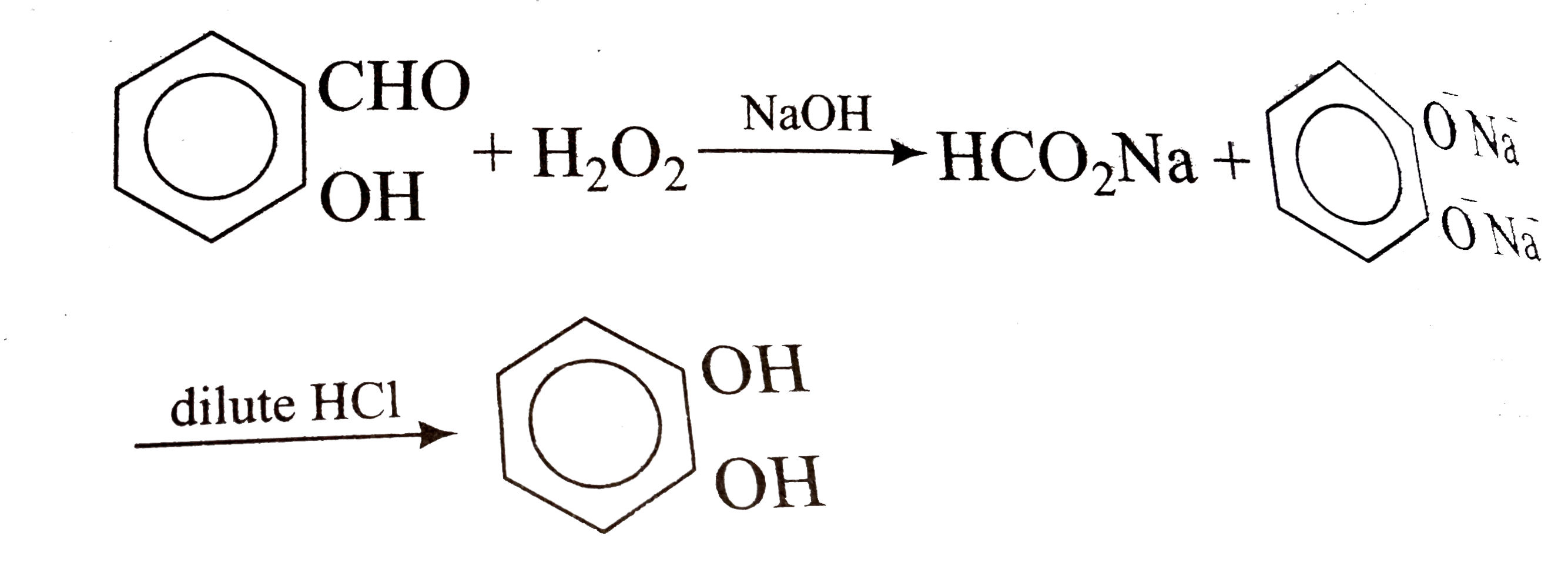Predicting the Autoaccelerating Hydrogen Peroxide Decomposition Rate after Mixing with Sodium Hydroxide | Industrial & Engineering Chemistry Research

Predicting the Autoaccelerating Hydrogen Peroxide Decomposition Rate after Mixing with Sodium Hydroxide | Industrial & Engineering Chemistry Research
![Oxidation in vitro of chromium(III) dietary supplements mer-[Cr(pic)3] and trans(S,S)-[Cr(Cys)2]− by hydrogen peroxide | Transition Metal Chemistry Oxidation in vitro of chromium(III) dietary supplements mer-[Cr(pic)3] and trans(S,S)-[Cr(Cys)2]− by hydrogen peroxide | Transition Metal Chemistry](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs11243-017-0168-2/MediaObjects/11243_2017_168_Sch1_HTML.gif)
Oxidation in vitro of chromium(III) dietary supplements mer-[Cr(pic)3] and trans(S,S)-[Cr(Cys)2]− by hydrogen peroxide | Transition Metal Chemistry

Predicting the Autoaccelerating Hydrogen Peroxide Decomposition Rate after Mixing with Sodium Hydroxide | Industrial & Engineering Chemistry Research
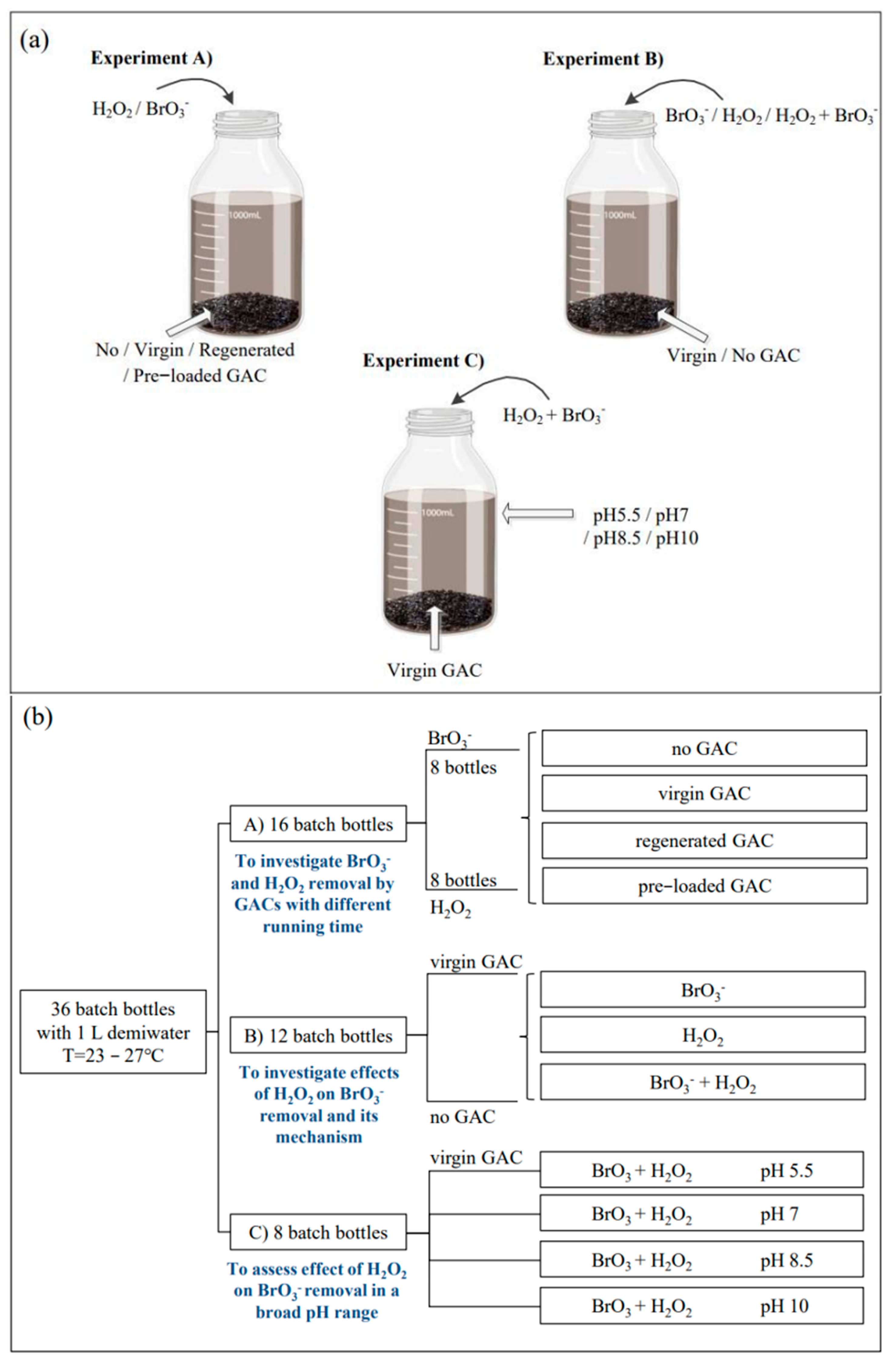
Water | Free Full-Text | Removal of Hydrogen Peroxide Residuals and By-Product Bromate from Advanced Oxidation Processes by Granular Activated Carbon

Predicting the Autoaccelerating Hydrogen Peroxide Decomposition Rate after Mixing with Sodium Hydroxide | Industrial & Engineering Chemistry Research

Relationship between structure and catalyst effect in the degradation kinetics of reactive dyes with hydrogen peroxide: UV-light versus sodium hydroxide - ScienceDirect

What happens when:Chromium hydroxide is treated with hydrogen peroxide in the presence of sodium hydroxide.
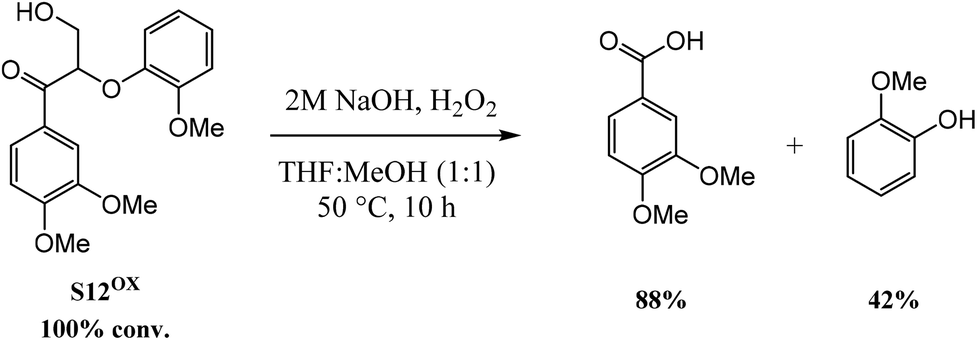
Hydrogen peroxide as an oxidant in biomass-to-chemical processes of industrial interest - Green Chemistry (RSC Publishing) DOI:10.1039/C9GC02445J