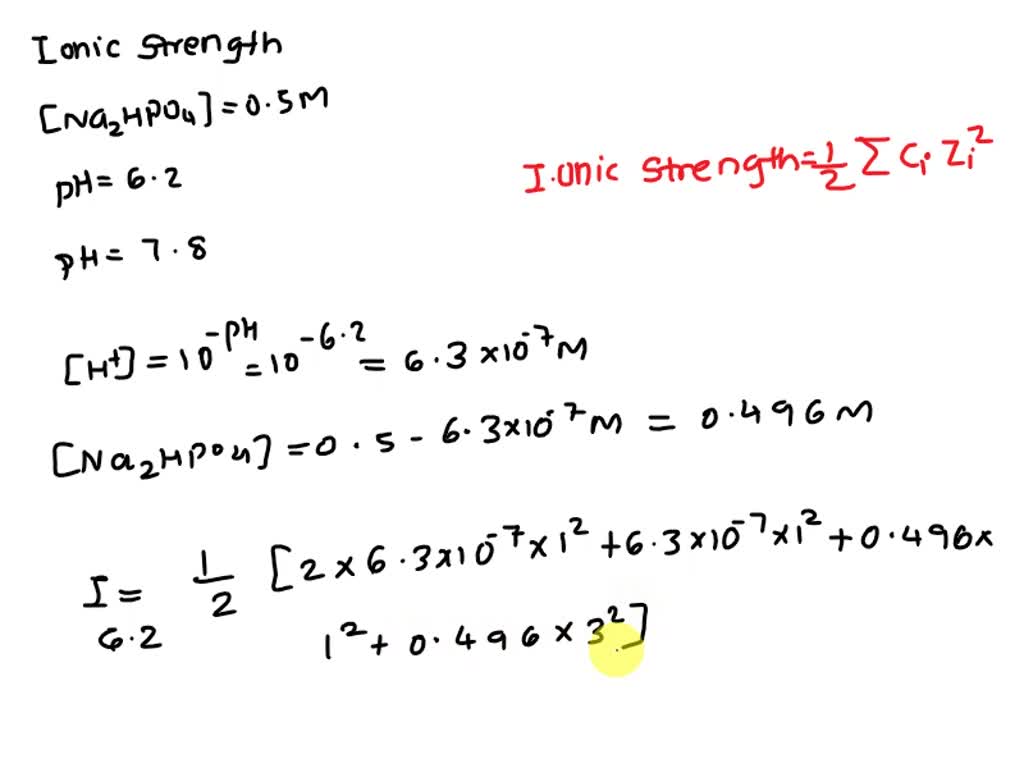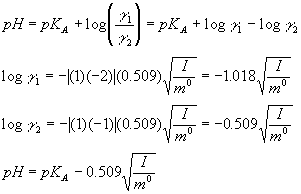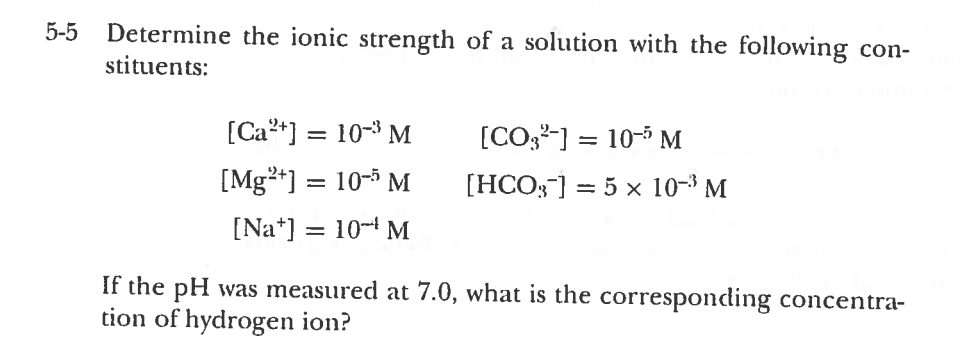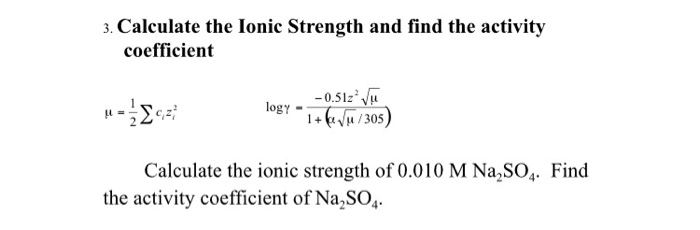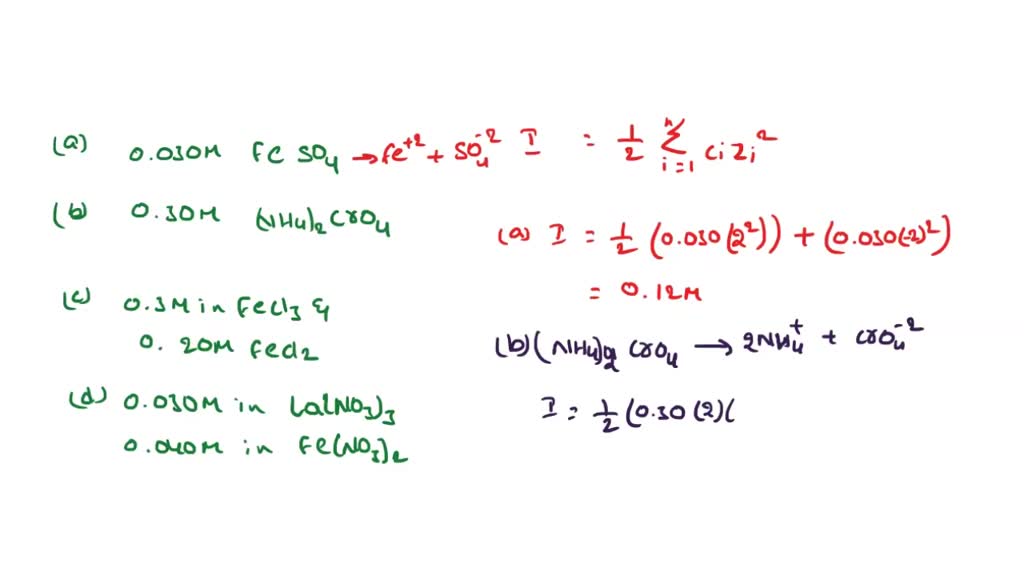
SOLVED: Calculate the ionic strength of a solution that is a. 0.030 M in FeSO4. Ionic strength b. 0.30 Min (NH)z Cr O4. Ionic strength c. 0.30 M in FeCls and 0.20
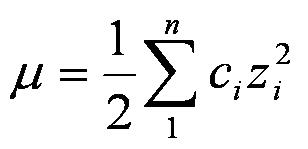
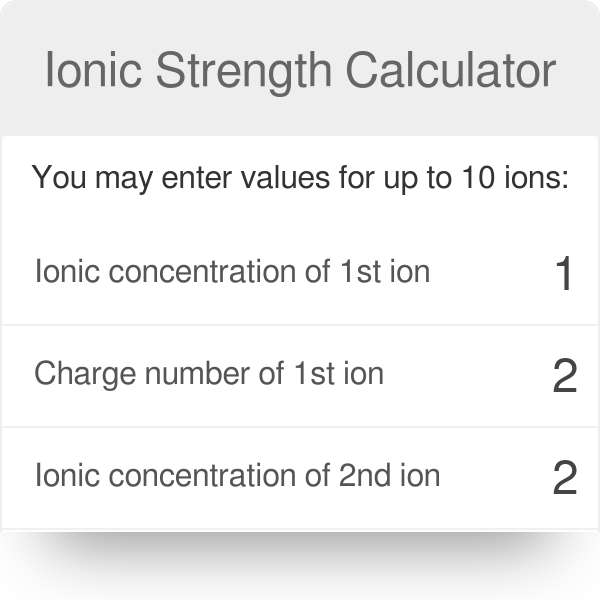
pH calculations and more in fundamentals of pharmaceutics. : What is ionic strength of solutions and how is it calculated?

25.4 g of iodine and 14.2 g of chlorine are made to react completely to yield a mixture of ICl and ICl3 . Calculate the number of moles of ICl and ICl3 formed.

Ionic strength - Introduction + total Explanation & formulas•MSc CHEMISTRY PHYSICAL• #notes - YouTube

Calculation of Ionic strength||How to calculate ionic strength ||Rank booster-3||Cpet-2021|| - YouTube

Ionic strength is sometimes stated as having units of molal (or molar) and other times stated as being unitless, depending on the book you read. The easiest. - ppt download