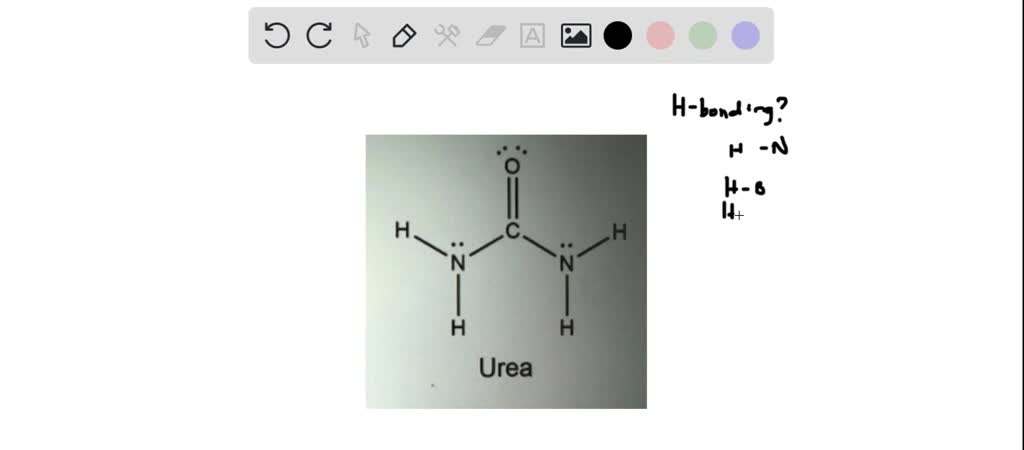
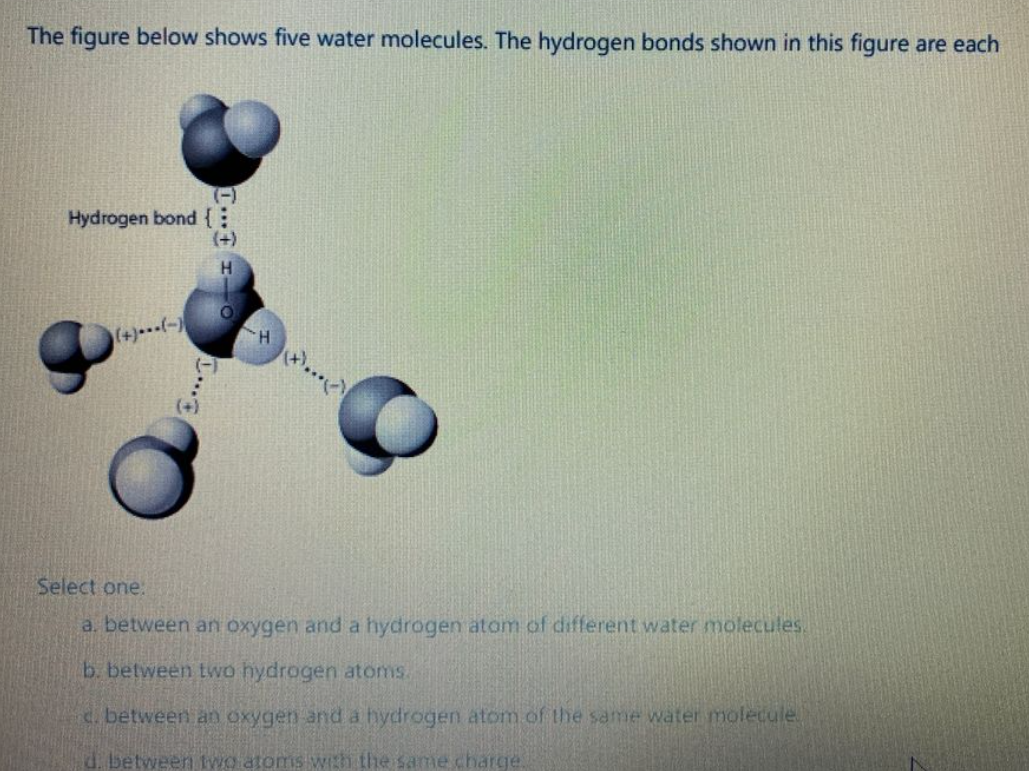
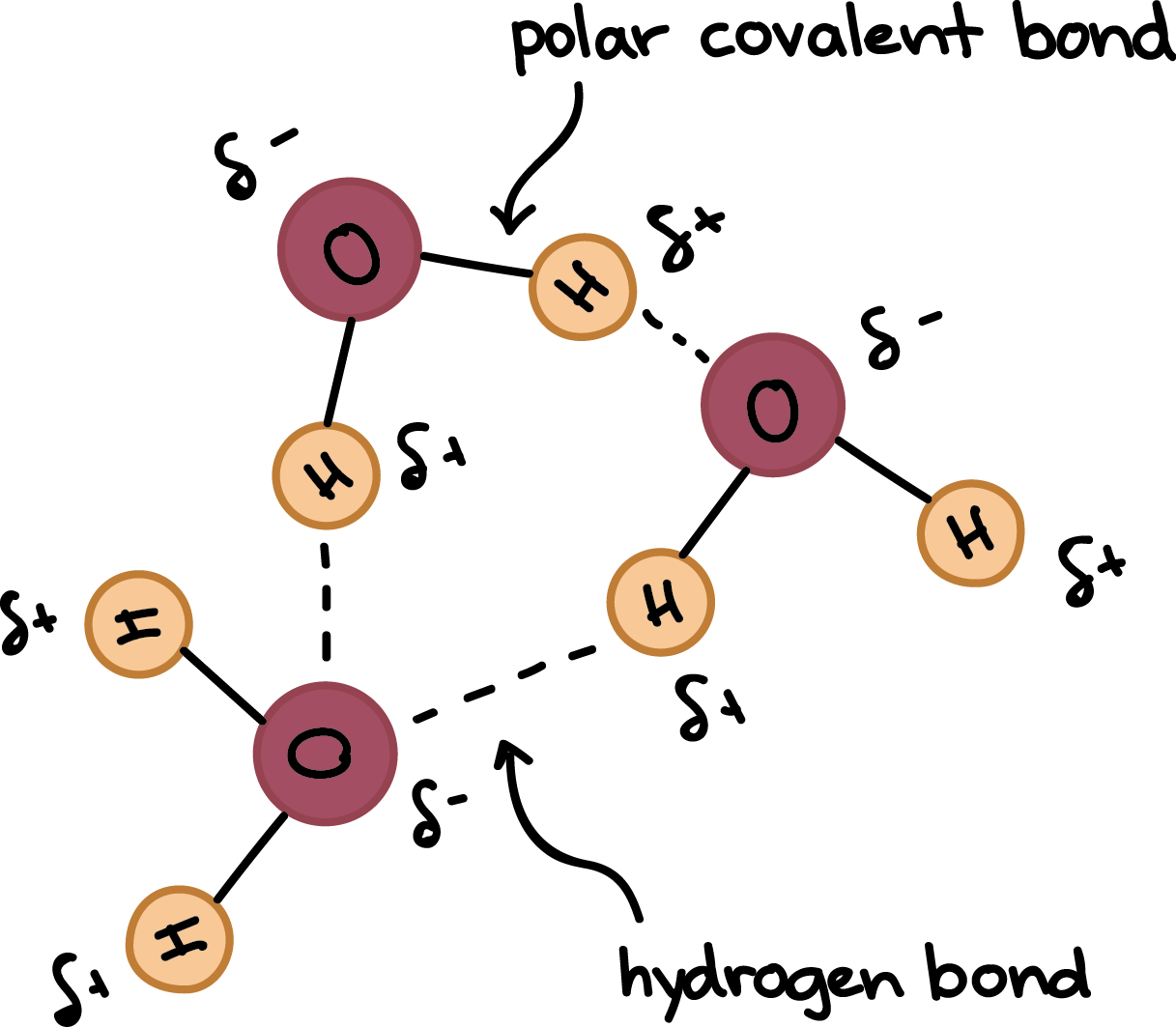What is the maximum theoretical number of water molecules that one urea molecule can hydrogen bond with? | Homework.Study.com

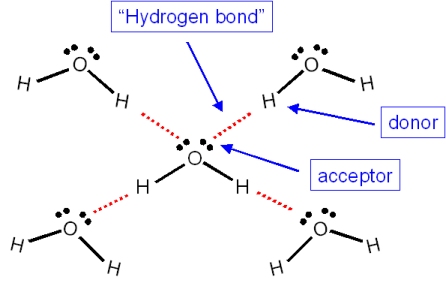
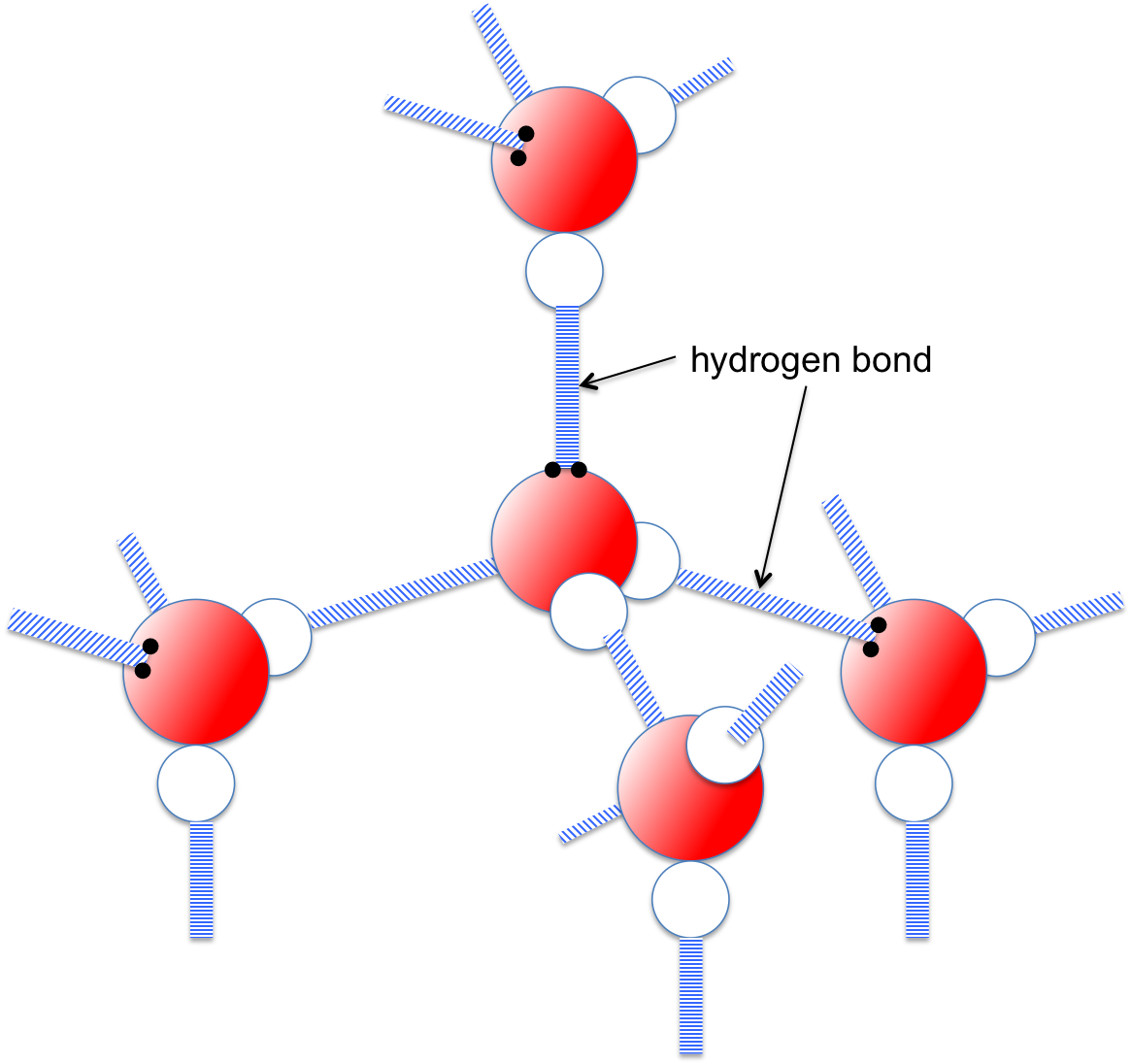
Each water molecule is joined to _____ other water molecules by ____ bonds. each water molecule is - Brainly.com
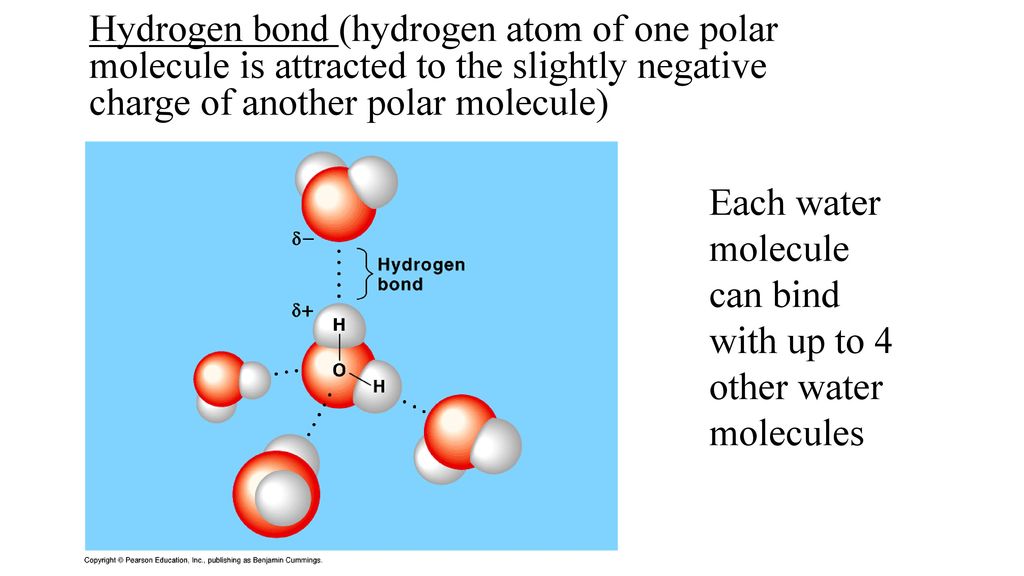
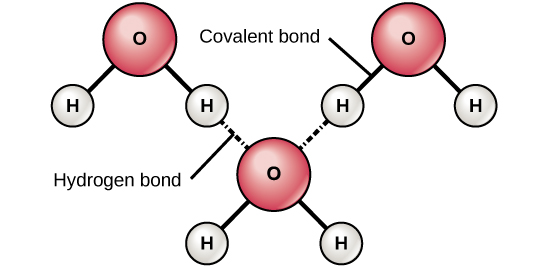
Both water and methanol have anomalously high boiling points due to hydrogen bonding, but the boiling point of water is greater than that of methanol despite its lower molecular mass. Why? -