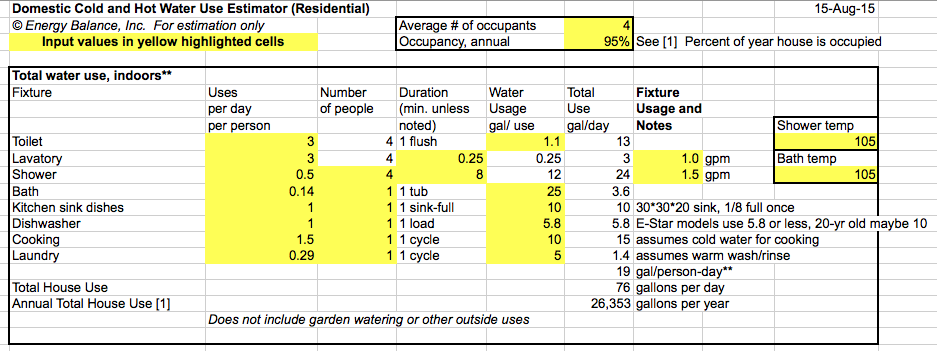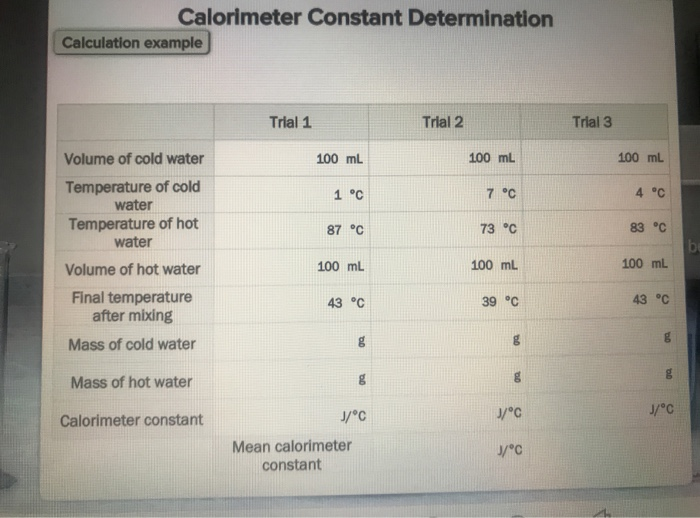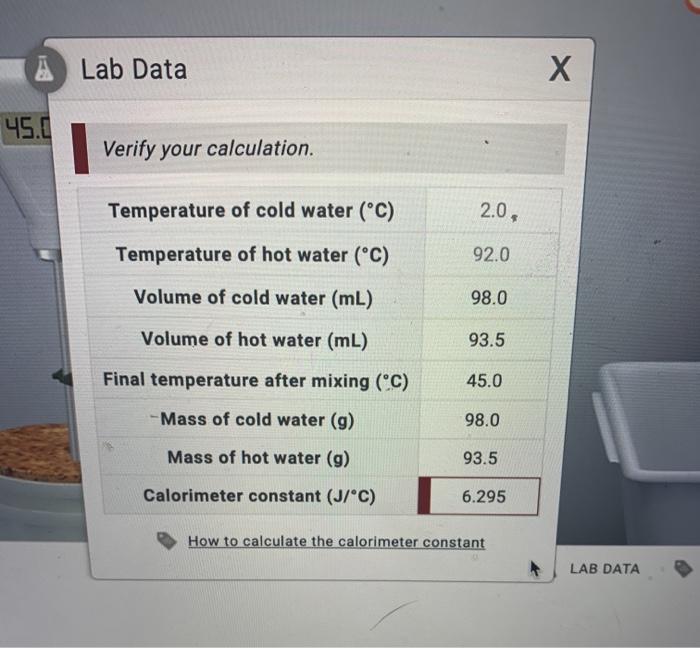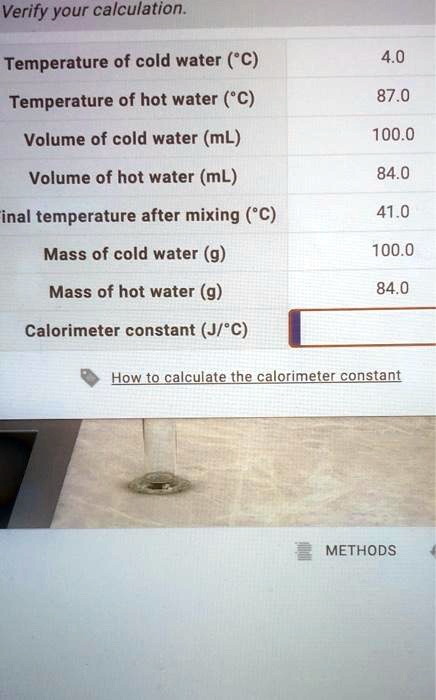
SOLVED: Verify your calculation: Temperature of cold water ("C) Temperature of hot water ("C) Volume of cold water (mL) 4.0 87.0 100.0 Volume of hot water (mL) 84.0 inal temperature after mixing ('

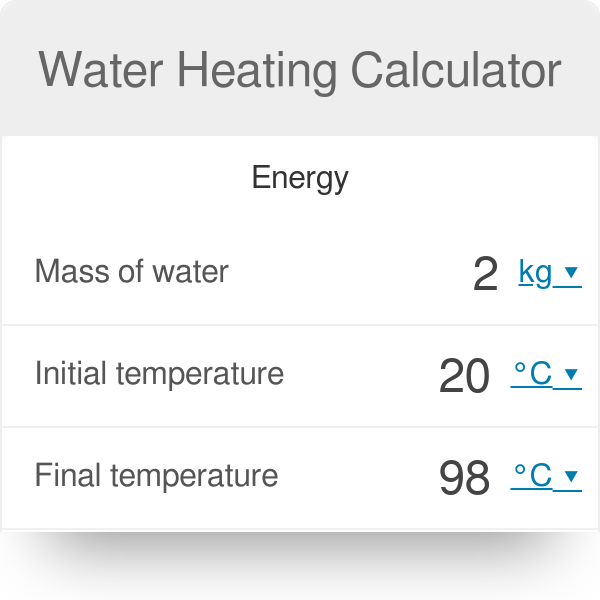
200 g of hot water at 80∘ C is added to 300 g of cold water at 10∘ C. Calculate the final temperature of the mixture of water. Consider the heat taken

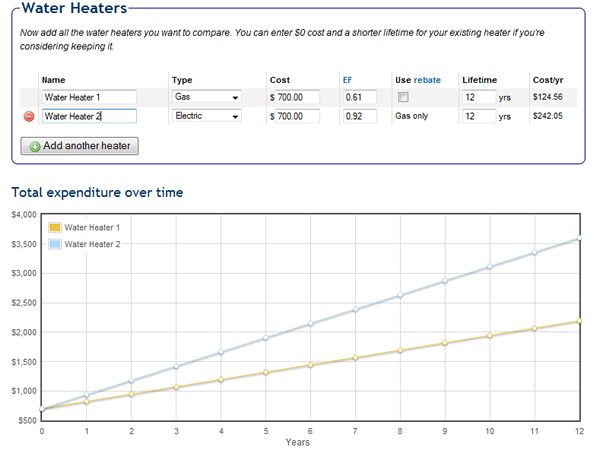
ratio - Equation to calculate percentage of $\color{red}{\text{hot}}$ water needed to raise $\color{blue}{\text{cold}}$ water to $x^\circ$ using mixing valve - Mathematics Stack Exchange

200 g of hot water at 80^oC is added to 300 g of cold water at 10^oC . Neglecting the heat taken by the container, calculate the final temperature of the mixture
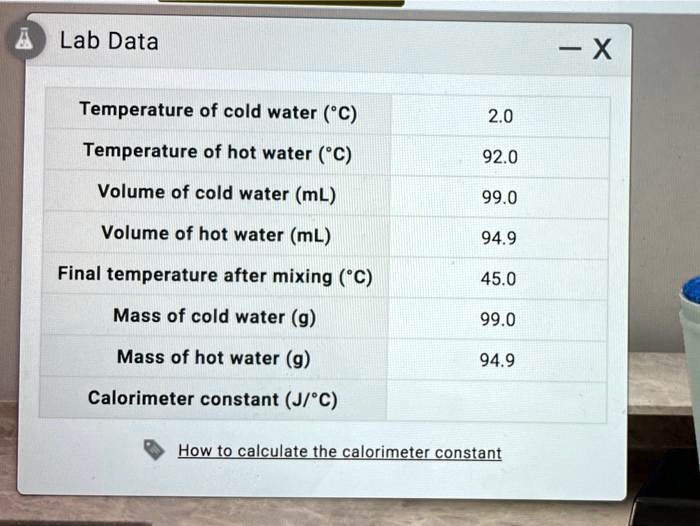
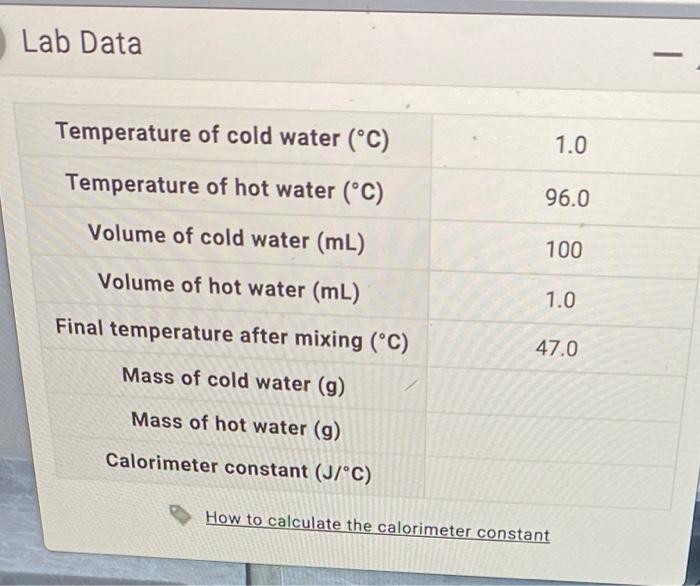
SOLVED: Lab Data X Temperature of cold water ('C) Temperature of hot water (C) Volume of cold water (mL) Volume of hot water (mL) Final temperature after mixing ("C) Mass of cold

200 g of hot water at 80^0 C is added to 300 g of cold water at 10^0 C. Neglecting the heat taken by the container, calculate the final temperature of the

200 g of hot water at 80^@C is added to 300 g of cold water at 10^@C. Neglecting the heat taken by the container, calculate the final temperature of the mixture of

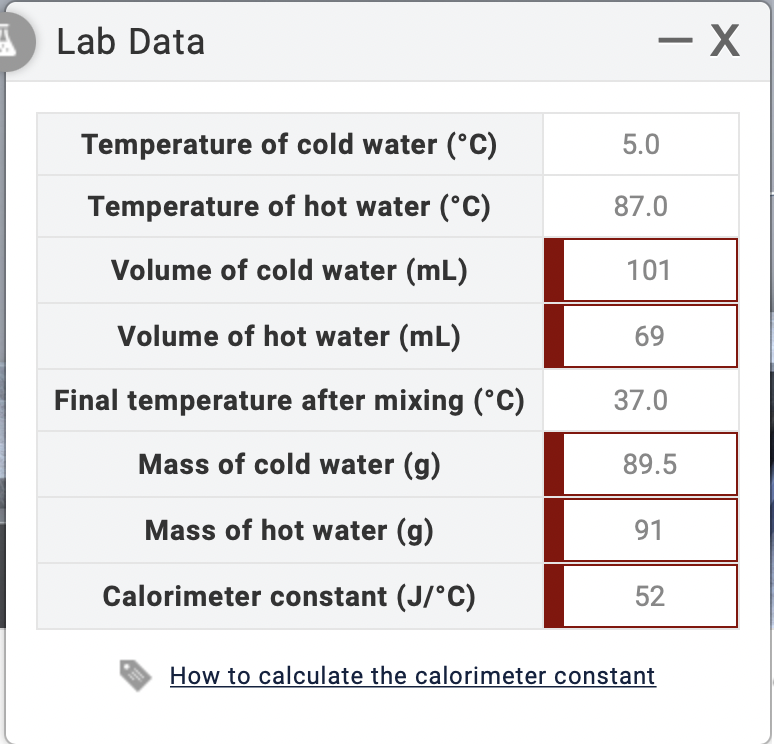
SOLVED: Lab Data X Verify your calculation. Did you report your data to the correct number of significant figures? Temperature of cold water (PC) 2.0 Temperature of hot water (PC) 92.0 Volume