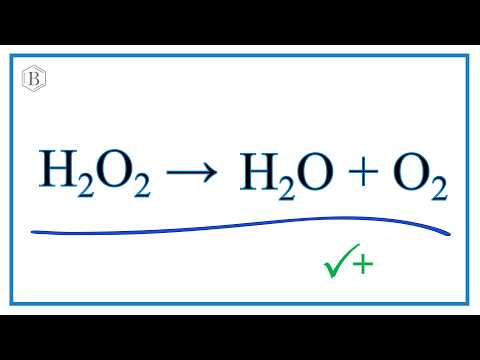
Clicker #1 Which of the following shows the correctly balanced equation for this reaction: H2O2 → H2O + O2 A) 3 H2O2 → 3 H2O + O2 B) H2O2 → 2 H2O + O2. - ppt download

H2O2=H2O+O2 balance the chemical equation @mydocumentary838. h2o2=h2o+o2 balance the equation. - YouTube

Reduction of O2 to H2O and its free radical intermediates (A) Lewis... | Download Scientific Diagram

If x, y, z are real numbers such that √(x - 1) + √(y - 2) + √(z - 3) = 0 then the values of x, y,z are respectively

Hydrogen peroxide generation from O2 electroreduction for environmental remediation: A state-of-the-art review - ScienceDirect
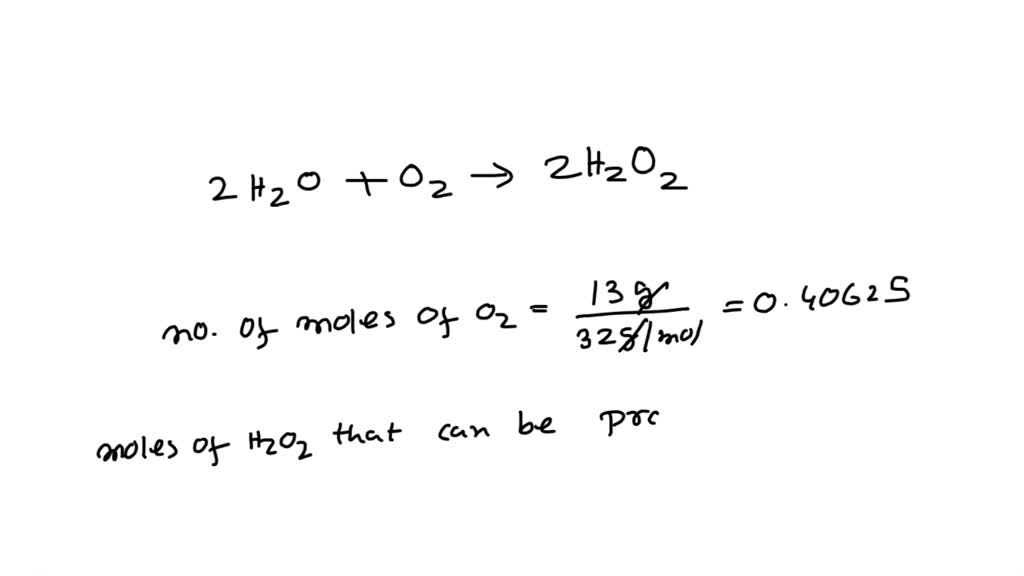
SOLVED: How many moles of H2O2 are produced in the production of hydrogen peroxide when 13 grams of oxygen are used?2 H2O+O2→2 H2O2 81 g H2O2 6.5 mol H2O2 0.81 mol H2O2 13 mol H2O2