
Buildings | Free Full-Text | Strength, Chloride Ion Penetration, and Nanoscale Characteristics of Concrete Prepared with Nano-Silica Slurry Pre-Coated Recycled Aggregate

Analysis of Operation Conditions of Ca(OH)2 Entrained Carbonator Reactors for CO2 Capture in Backup Power Plants | ACS Omega

Effect of Impurities on the Decarbonization of Calcium Carbonate Using Aqueous Sodium Hydroxide | ACS Sustainable Chemistry & Engineering
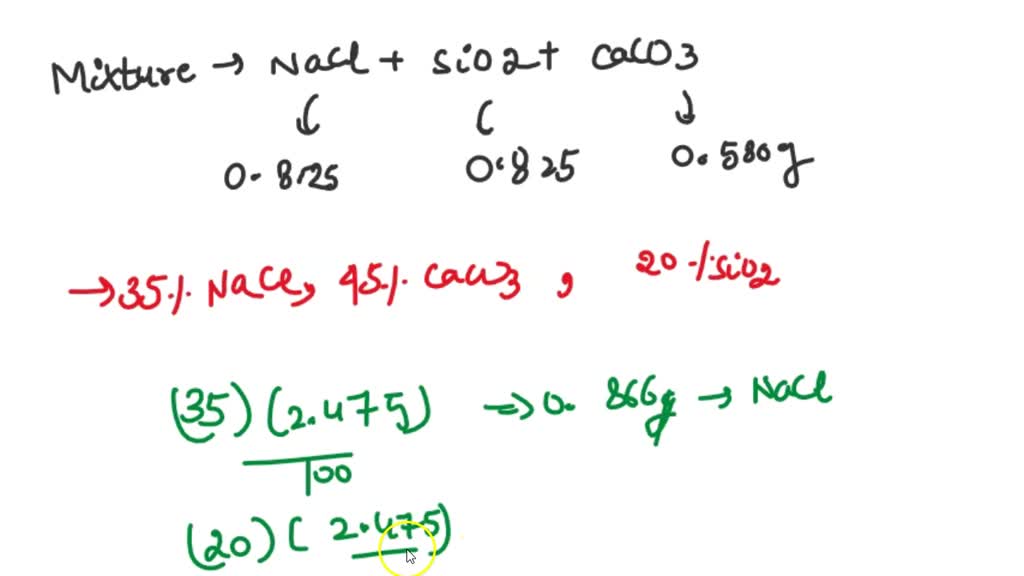
SOLVED: You analyzed 2.475 g of a mixture of NaCl, SiO2, and CaCO3. You isolate 0.825 g NaCl, 0.825 g of CaCO3, and 0.580 g SiO2. You were told that the original
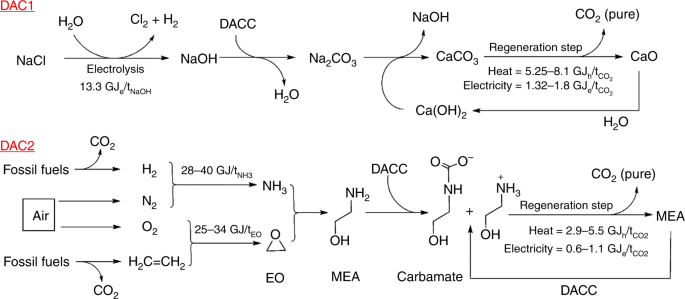
Unrealistic energy and materials requirement for direct air capture in deep mitigation pathways | Nature Communications

Effect of Impurities on the Decarbonization of Calcium Carbonate Using Aqueous Sodium Hydroxide | ACS Sustainable Chemistry & Engineering
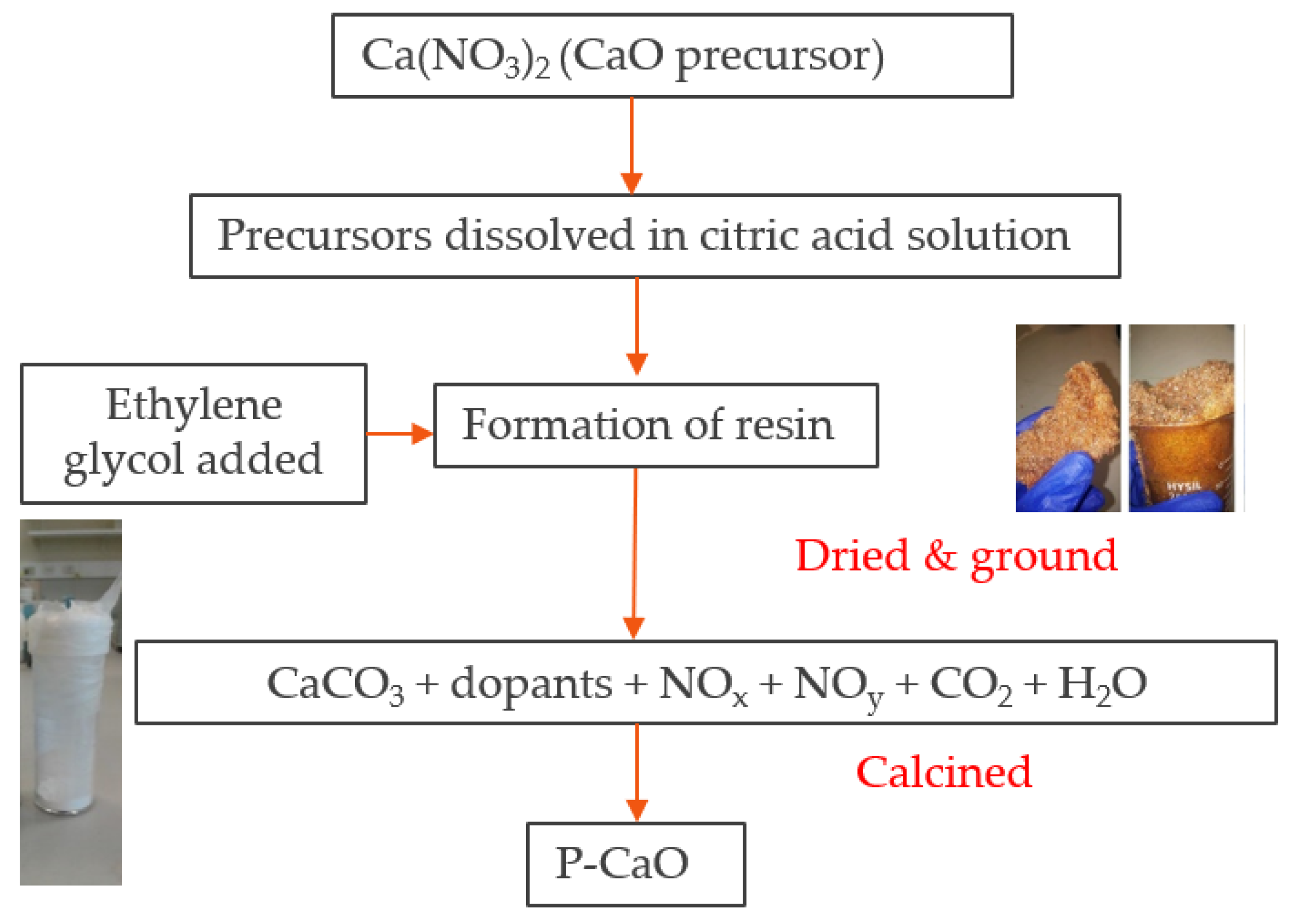
Applied Sciences | Free Full-Text | Comparative Kinetic Analysis of CaCO3/CaO Reaction System for Energy Storage and Carbon Capture
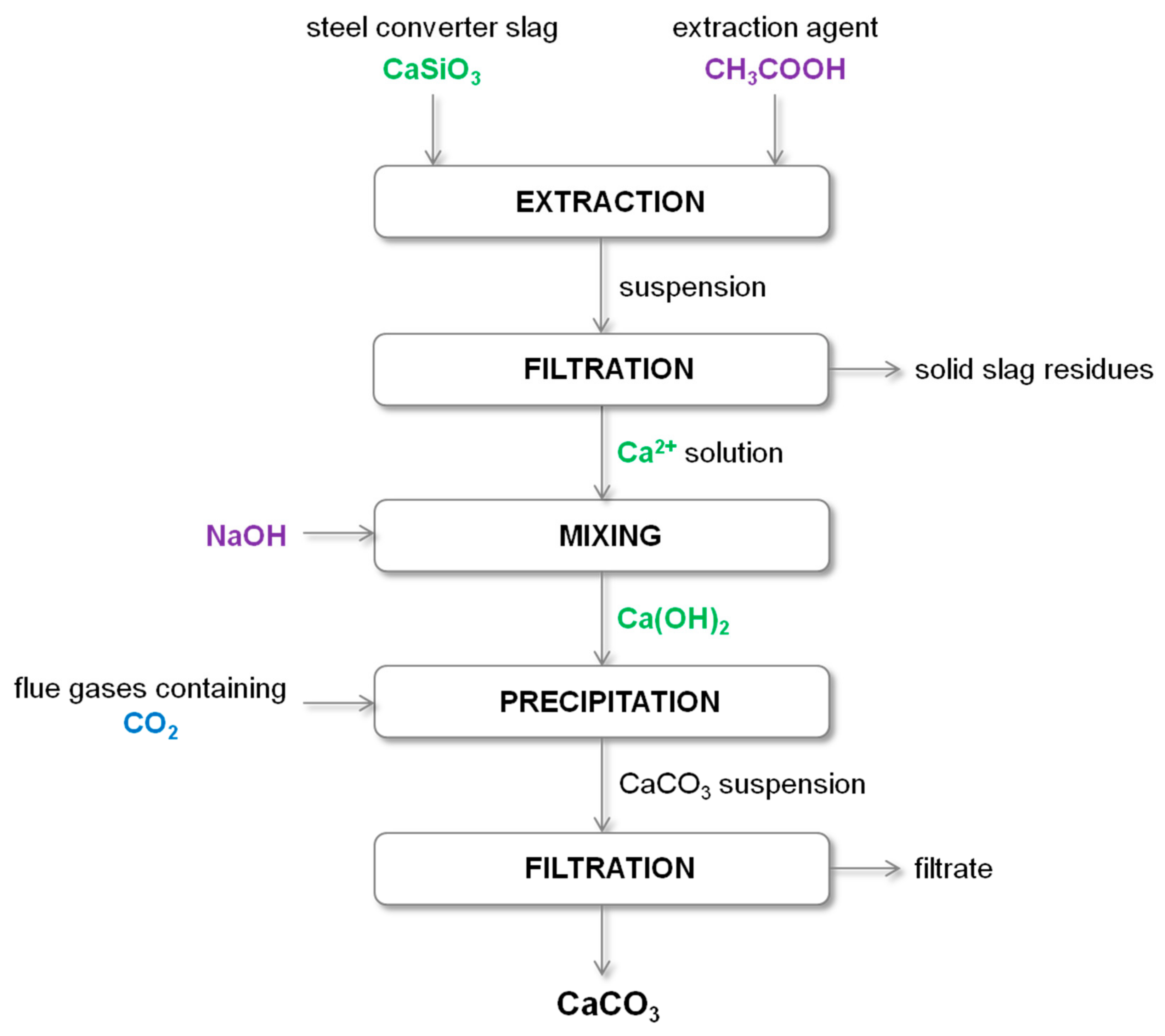
Energies | Free Full-Text | Utilization of Gaseous Carbon Dioxide and Industrial Ca-Rich Waste for Calcium Carbonate Precipitation: A Review

Electrochemical Splitting of Calcium Carbonate to Increase Solution Alkalinity: Implications for Mitigation of Carbon Dioxide and Ocean Acidity | Environmental Science & Technology
Relating Structural and Microstructural Evolution to the Reactivity of Cellulose and Lignin during Alkaline Thermal Treatment wi

Analysis of Operation Conditions of Ca(OH)2 Entrained Carbonator Reactors for CO2 Capture in Backup Power Plants | ACS Omega

Calcium-Looping Performance of Biomineralized CaCO3 for CO2 Capture and Thermochemical Energy Storage | Industrial & Engineering Chemistry Research

SOLVED: Enter the equation for precipitate formation in the test tube containing the calcium hydroxide solution

Polymorph Selection and Structure Evolution of CaCO3 Mesocrystals under Control of Poly(sodium 4-styrenesulfonate): Synergetic Effect of Temperature and Mixed Solvent | Crystal Growth & Design

Encapsulation of Proteins into CaCO3 by Phase Transition from Vaterite to Calcite | Crystal Growth & Design







