
Calculate the work done during isothermal reversible expansion of one mole ideal gas from 10 atm to 1 atm at 300 K.

Calculate the volume occupied by 8.8g of CO2 at 31.1C and 1 bar pressure. R= 0.083 bar dm3 K-1 mol-1 - YouTube

Convert 22 g of carbon dioxide `(CO_(2))` into moles. (Atomic masses : `C = 12 u, O = 16 u`) - YouTube

A Step toward the Quantification of Noncovalent Interactions in Large Biological Systems: The Independent Gradient Model-Extremely Localized Molecular Orbital Approach | Journal of Chemical Information and Modeling

Metal–CO2 Electrochemistry: From CO2 Recycling to Energy Storage - Wang - 2021 - Advanced Energy Materials - Wiley Online Library

Calculate mass of co2 produced by combustion of 8g of methane - Chemistry - Some Basic Concepts of Chemistry - 13818719 | Meritnation.com

Calculate the volume occupied by 8.8 g of CO 2 at 31.1 ^∘C and 1 bar pressure. (R = 0.083 bar L K ^-1 mol ^-1 )

The maximum work done in expanding 16g oxygen at 300K and occupying a volume of 5dm^3 isothermally until the volume become 25dm^3 is:
How to calculate the work done if three moles of an ideal gas are compressed reversibly and isothermally from a volume of 10 litres to 5 litres - Quora
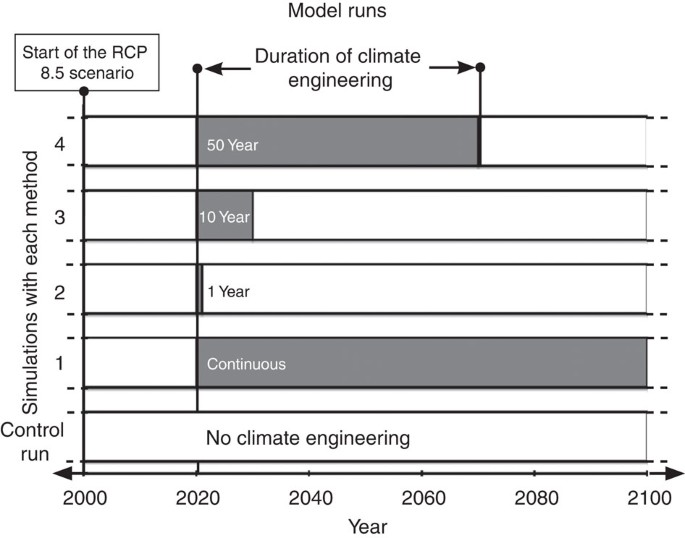
Potential climate engineering effectiveness and side effects during a high carbon dioxide-emission scenario | Nature Communications

An Accurate and Efficient Look-up Table Equation of State for Two-Phase Compressible Flow Simulations of Carbon Dioxide | Industrial & Engineering Chemistry Research

Basic Principles and Calculations in Chemical Engineering 32e14f234b | PDF | Units Of Measurement | Mass

PDF) Oxygen isotope anomaly in tropospheric CO2 and implications for CO2 residence time in the atmosphere and gross primary productivity

what volume of `CO_2` will be liberated at STP if 12 g of carbon is burnt in excess of oxygen ? - YouTube

ACT Practice Questions.docx - ACT Practice Questions A scientist studying hemoglobin investigated the impact of temperature and carbon dioxide CO2 | Course Hero









